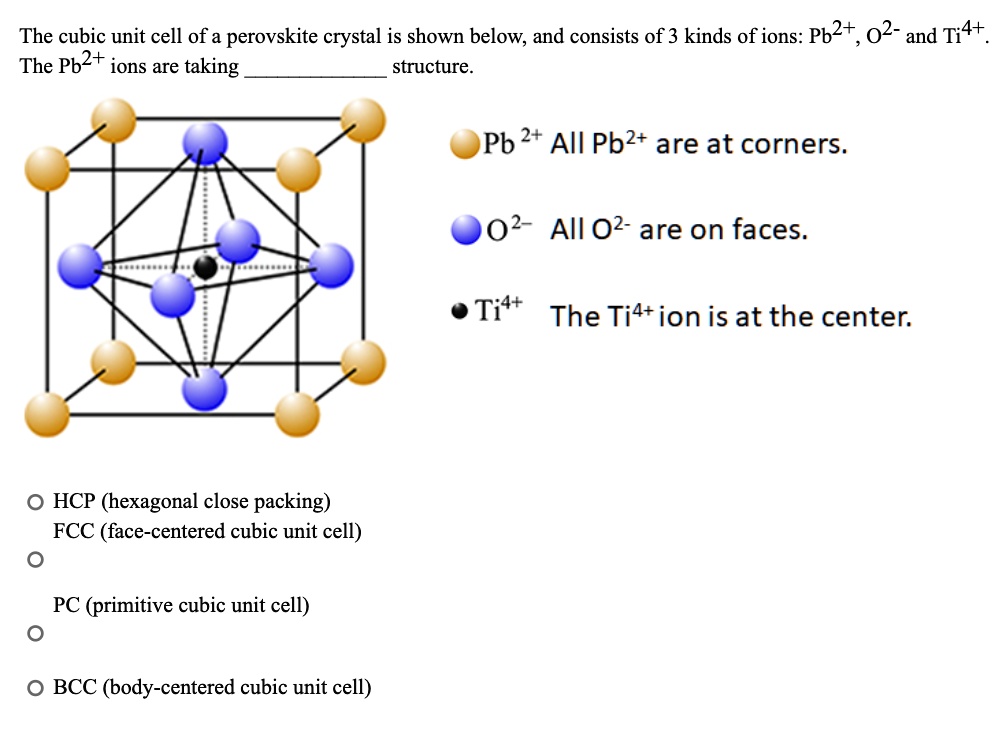
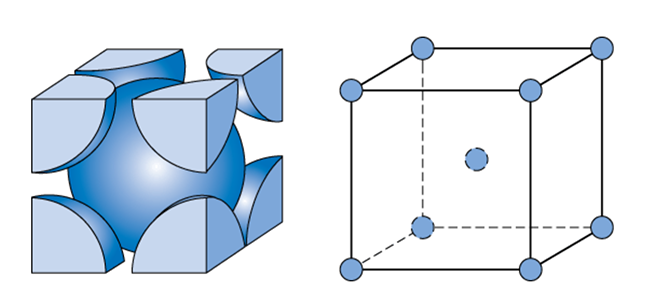
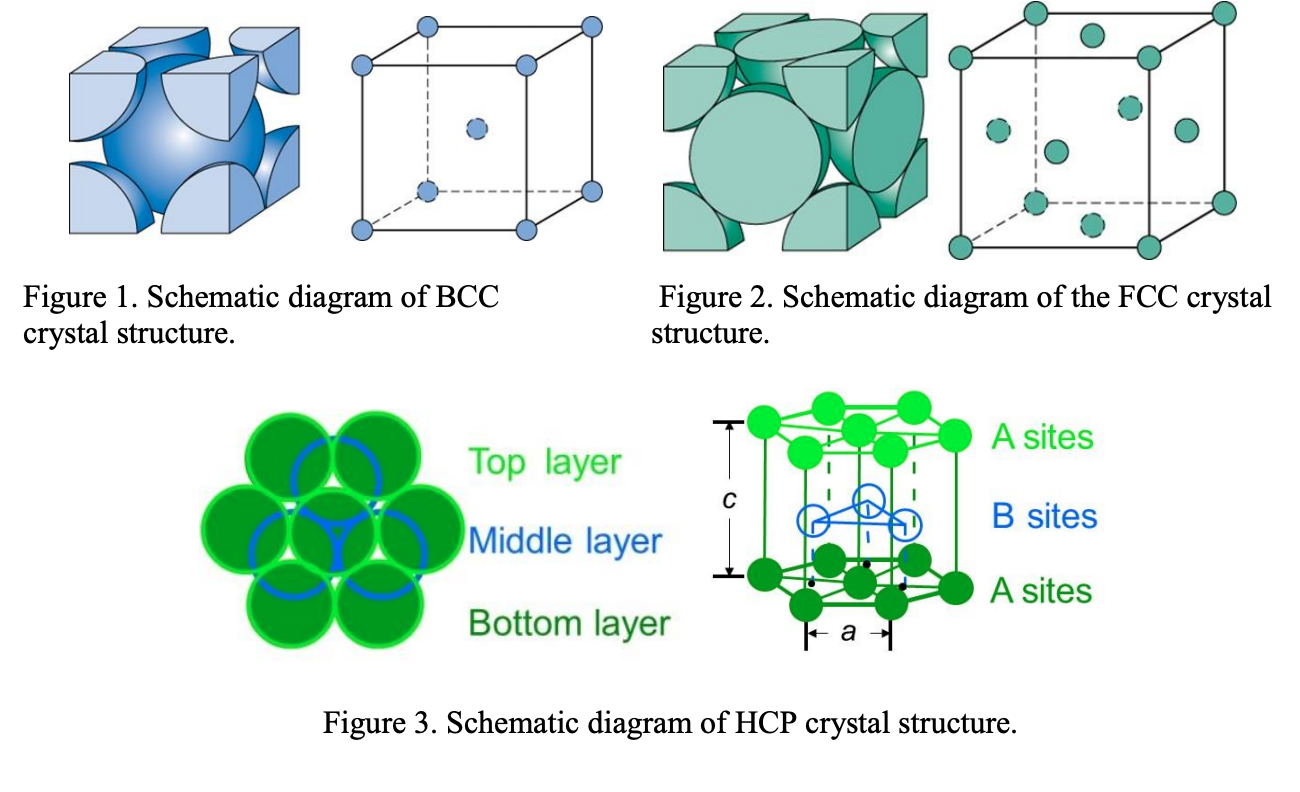
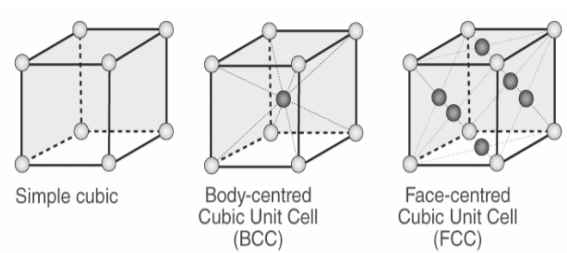
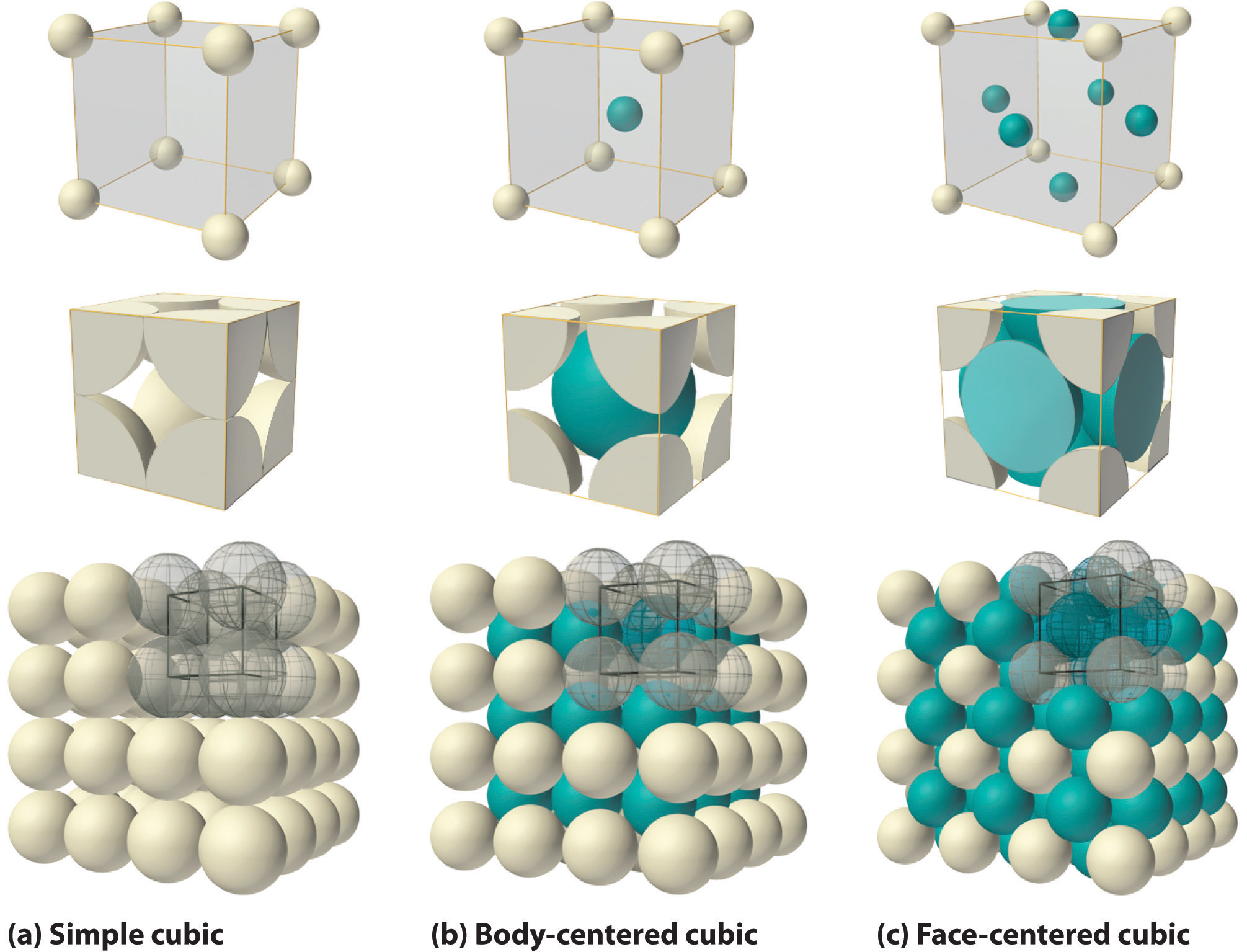
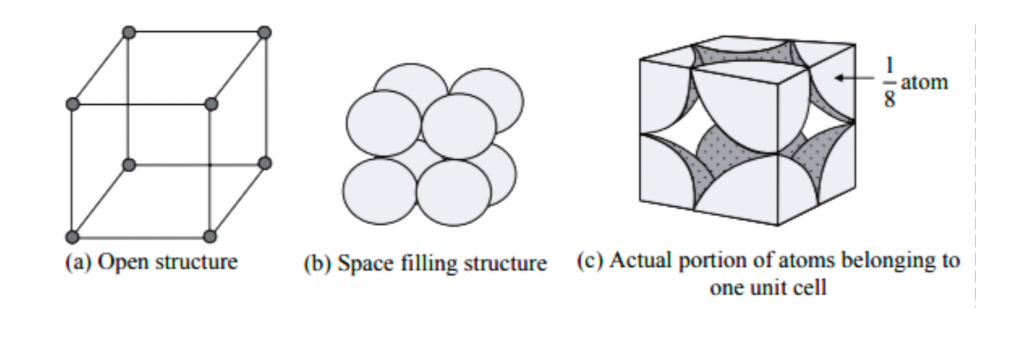
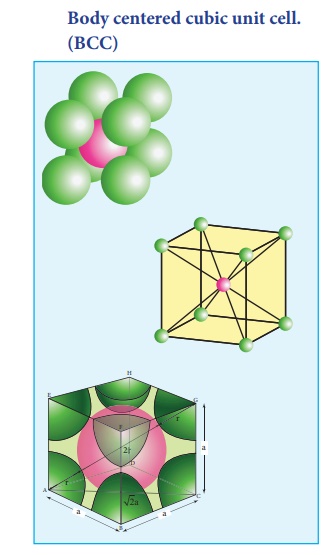
What Is the Difference Between FCC and BCC? (Crystal Structure, Properties, Interstitial Sites, and Examples) – Materials Science & Engineering

SOLVED: Title: Face Centered Unit Cell Fig: Face-Centered Cubic (FCC) Unit Cell Ball-and-Stick Model Space-filled Model Stacking: How many unit cells share each atom on each face of the face-centered cubic unit

What is a Unit Cell? - Definition, Types of Unit Cell, Primitive Unit Cell, BCC & FCC, Volume of HCP Unit Cell
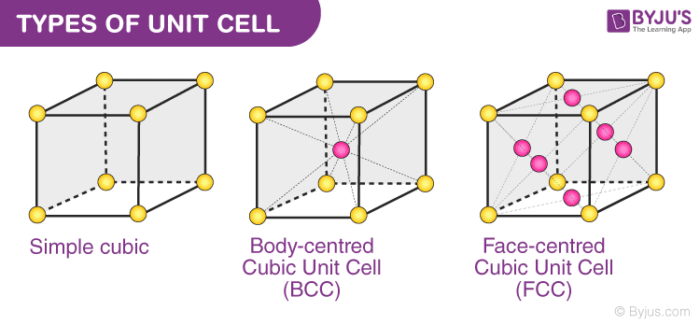
What is a Unit Cell? - Definition, Types of Unit Cell, Primitive Unit Cell, BCC & FCC, Volume of HCP Unit Cell
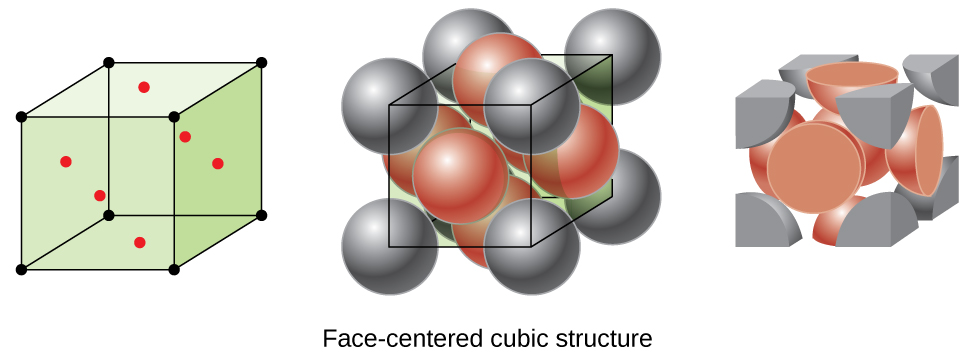
Types of Unit Cells: Body-Centered Cubic and Face-Centered Cubic (M11Q5) – UW-Madison Chemistry 103/104 Resource Book
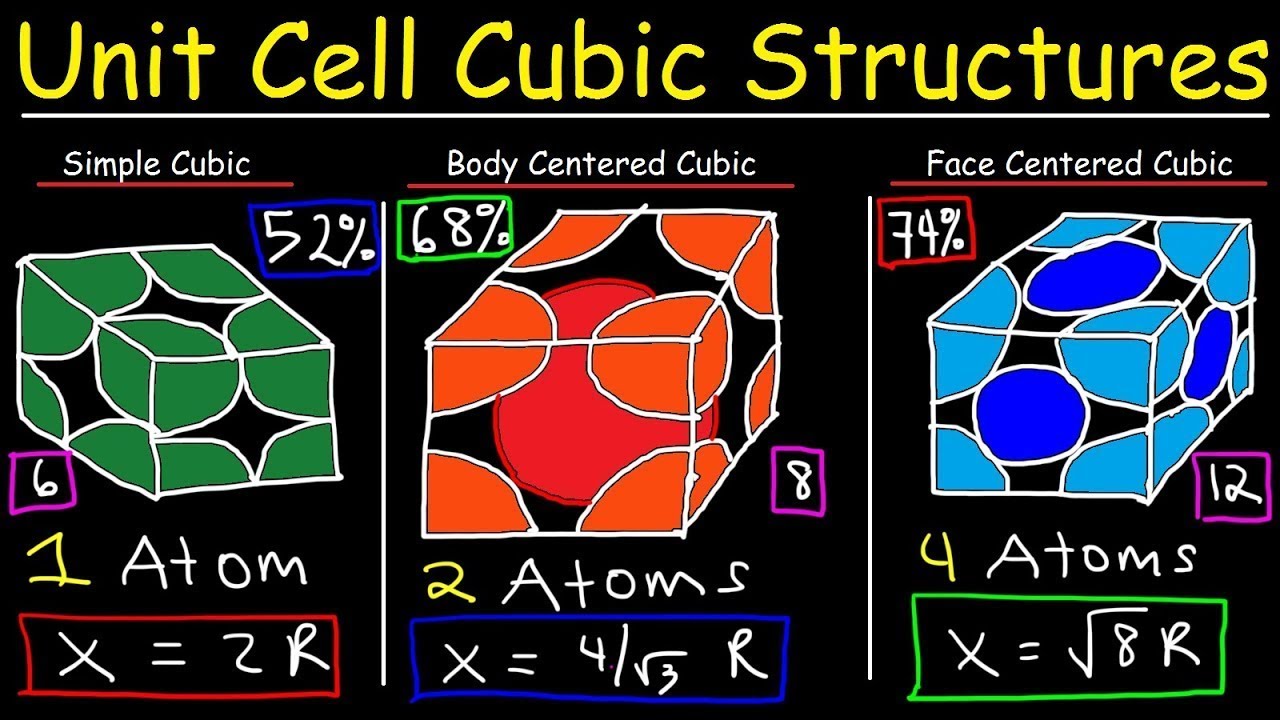
Unit Cell Chemistry Simple Cubic, Body Centered Cubic, Face Centered Cubic Crystal Lattice Structu - YouTube